 You will see in the next chapters that the chemical properties of elements are determined by the number of valence electrons. The table is arranged such that elements that have the same number of valence electrons fall under the same group. We can see from the electron configuration of a carbon atom-1 s 22 s 22 p 2-that it has 4 valence electrons (2 s 22 p 2) and 2 core electrons (1 s 2). Explanation: The periodic table indicates the number of valence electrons an element has through its arrangement into groups and periods. Answer: All have an ns2np5 electron configuration, one electron short of a noble gas electron configuration. Use the periodic table to predict the characteristic valence electron configuration of the halogens in group 17. 1: Periodic table by Dmitri Mendeleev, 1871. By extrapolation, we expect all the group 2 elements to have an ns2 electron configuration. After the development of quantum mechanics, it was shown that the halogens all had seven valence electrons, supporting their original placement into the same group on Mendeleev's periodic table. For a fully interactive version with orbitals, isotopes, compounds, and free printouts, visit. As a gas or vapor, the halogens all had a pungent odor. Valence shell electrons (or, more simply, the valence electrons) are the electrons in the highest-numbered shell, or valence shell, while core electrons are the electrons in lower-numbered shells. Design Copyright © 2017 Michael Dayah (). Electron configuration chart of all Elements is mentioned in the table below. 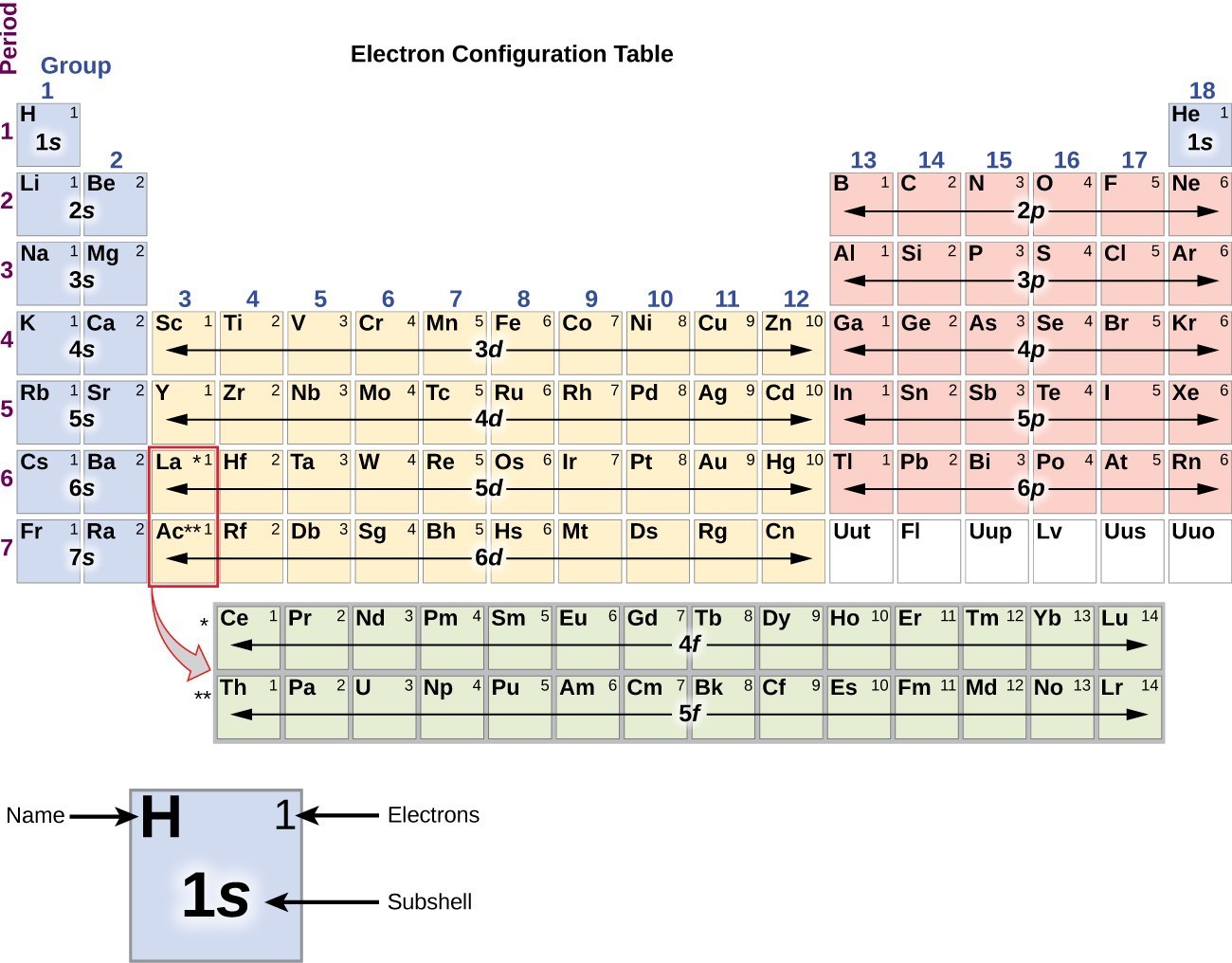
It is the transition metal due to which it has the variable valency. The valency of gold may be either of 1 or 3 in different scenarios. The interaction of valence electrons helps in understanding the chemical binding of elements. Thus, it is convenient to separate electrons into two groups. It will further provide the accurate numbers of valence electrons of atoms. This periodic table in PDF format can be downloaded from here. This table also contains the element number, element symbol, element name and atomic weights of each element. The path a specific element will take depends on where the electrons are in the atom and how many there are. This color periodic table contains the most common valence charges of the elements. 
The electron configurations are written in the noble gas notation. This downloadable color periodic table contains each elements atomic number, atomic mass, symbol, name, and electron configuration. \,3s^1 \nonumber \]Ī chemical reaction results from electron removal, electron addition, or electron sharing of the valence electrons of the different atoms. Color Periodic Table With Electron Configurations. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
